How Many Silicon 30 Atoms Are Found in This Plate
The mass of a silicon- 30 atom is 2997376 mathrmu and the percent isotopic abundance of silicon- 30 is 310. The mass of a silicon- 30 atom is 2997376 mathrmu and the percent natural abundance of silicon- 30 is 310.

How Many Silicon 30 Atoms Are Found In This Plate Socratic
Monel metal is a corrosion-resistant copper-nickel alloy used in the electronics industry.
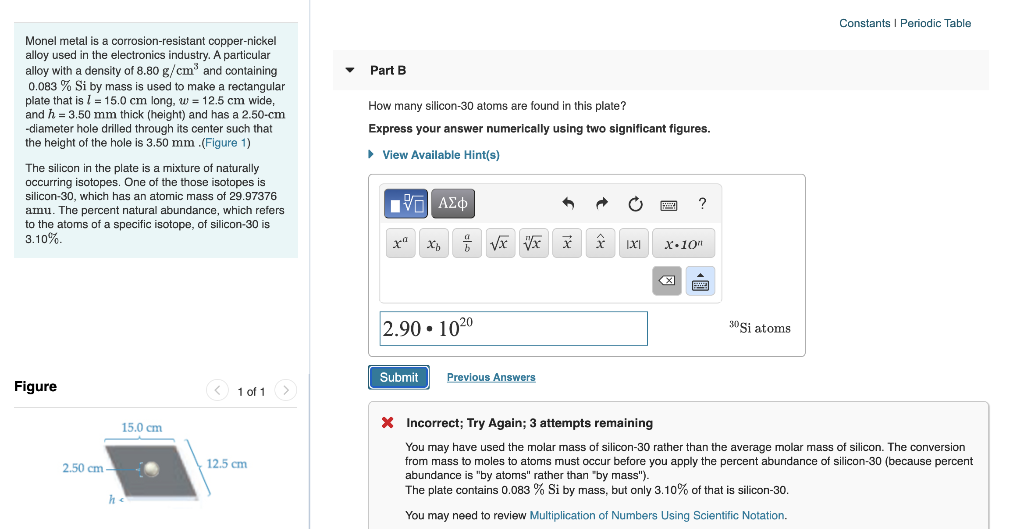
. Preciso de uma mensagem para uma amiga encontrista. Assuming that this is also the case for our plate the number of silicon-30 atoms will be. How many silicon-30 atoms are found in this plate.
310 31541021 atoms 0031 31541021 atoms 97781019 atoms. What is the volume of the plate. The silicon in the plate is a mixture of naturally occurring isotopes.
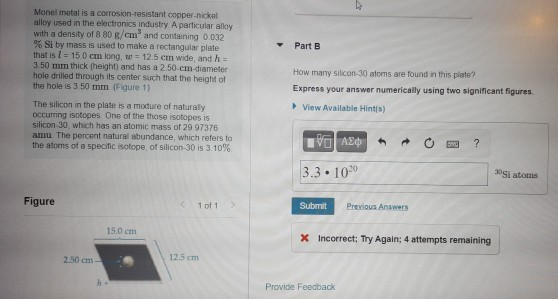
How many moles of silicon-30 atoms are found in the plate. How many silicon-30 atoms are found in this plate. The plate contains 0032 Si which means.
How many silicon-30 atoms are found in this plate. How many silicon-30 atoms are found in this plate. One of the those isotopes is silicon-30 which has an atomic mass of 2997376 a mu and a percent natural abundance of 310.
Carbon-12 contains 6 protons and 6 neutrons. How many silicon-30 atoms are found in this plate. The percent natural abundance of silicon-30 is 310.
The mass of a silicon - 30 atom is 2997376 u and the percent natural abundance of silicon - 30 is 310 Extra. Mass Si 5775 g x 000032. Very hard need help please.
One of the those isotopes is silicon-30 which has an atomic mass of 2997376 amu. One of the those isotopes is silicon-30 which has an atomic mass of 2997376 amu. Problem 83 Deuterium 2 mathrmH20140 mathrmu is sometimes used to replace the principal hydrogen isotope 1 mathrmH in chemical studies.
Mass Si 01848 g Si of Si atoms 01848 g 1 mol Si 2809 g si60221023 atoms1 mol Si of Si atoms 39611021 atoms Calculating the of 30 Si atoms at 310. Back to users profile. The percent natural abundance which refers to the atoms of a specific isotope of silicon-30 is 310.
How many silicon-30 atoms are found in this plate. 310 of these are Si-30. The percent natural abundance which refers to the atoms of a specific isotope of silicon-30 is 310.
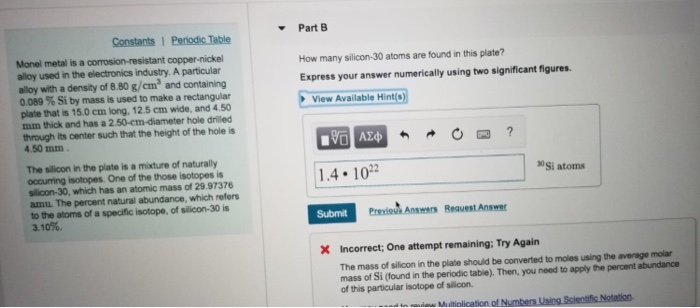
Express your answer numerically using two significant figures. The percent natural abundance which refers to the atoms of a specific isotope of silicon-30 is 310. The mass of a silicon-30 atom is 2997376 u and the percent isotopic abundance of silicon-30 is 310.
The least amount of significant figures in the question is 3 so our answer is going to have 3 significant figures. How many silicon- 30 atoms are found in this plate. What is the mass percentage of the gold in the alloy.
A particular alloy with a density of 88 gcm3 and containing 0078 Si by mass is used to make a rectangular plate 130 cm long 105 cm wide 50 mm thick and has a 225 cm diameter hole drilled through its center. One of the those isotopes is silicon-30 which has an atomic mass of 2997376 amu and a percent natural abundance of 310. Among students who drink what percentage typically have not drank in the last month.
What is the mass percentage of the palladium in the alloy. 84 An alloy that melts at about the boiling point of water has Bi Pb and Sn atoms in the ratio 1065 respectively. Up to 256 cash back The silicon in the plate is a mixture of naturally occurring isotopes.
003103794E18 atoms 1176E20 atoms of Si-30. How many silicon-30 atoms are found in this plate. Of 30Si atoms 39611021 atoms 000310 of 30Si atoms 12281019 atoms.
The radius of the nucleus is approximately 27 fm femtometers and the radius of the atom is approximately 70 pm picometers. How many silicon-30 atoms are found in this plate. Monel Metal Part B How many silicon-30 atoms are found in this plate.
017675 g1 mol280855 g6022E23 atoms1 mol 3794E21 atoms of Si. Soid use the average atomic mass of silicon and Avogadros number to find the number of silicon atoms. The mass of a silicon-30 atom is 2997376 u and the percent isotopic abundance of silicon-30 is 310.
How many silicon 30 atoms are found in this plate. The silicon in the plate is a mixture of naturally occurring isotopes. How many silicon-30 atoms are found in this plate.
How many silicon- 30 atoms are found in this plate. Up to 256 cash back The silicon in the plate is a mixture of naturally occurring isotopes. One of the those isotopes is silicon-30 which has an atomic mass of 2997376 amu.
The silicon in the plate is a mixture of naturally occurring isotopes. What is the volume of this plate B. The question tells us that 310of silicon atoms are of the silicon-30 isotope.
Solved Part B Monel Metal Is A Corrosion Resistant Chegg Com

Solved Constants Periodic Table Part B How Many Silicon 30 Chegg Com
Solved Part B How Many Silicon 30 Atoms Are Found In This Chegg Com

Comments
Post a Comment